Ahmedabad
(Head Office)Address : 506, 3rd EYE THREE (III), Opp. Induben Khakhrawala, Girish Cold Drink Cross Road, CG Road, Navrangpura, Ahmedabad, 380009.
Mobile : 8469231587 / 9586028957
Telephone : 079-40098991
E-mail: dics.upsc@gmail.com
CAR-T Cell Therapy
News: Recently, India’s first CAR-T cell therapy to receive approval is NexCAR19, developed by ImmunoACT. This therapy is a CD19-targeted CAR-T cell therapy product used to treat relapsed/refractory B-cell lymphomas and leukemia.
Background:
• The therapy was developed as a result of a decade-long collaborative effort between IITBombay and Tata Memorial Centre (TMC). The clinical trial, which involved 60 patients, showed an overall response rate of around 70%.
• The Central Drugs Standard Control Organization (CDSCO) has authorized the commercial launch of this therapy in India.
• ImmunoACT intends to make NexCAR19 available to its partner hospitals as soon as possible. The therapy is expected to be priced
in the Rs 30-40 lakh range per dose, making it significantly more affordable compared to existing treatments.
What is CAR-T cell therapy?
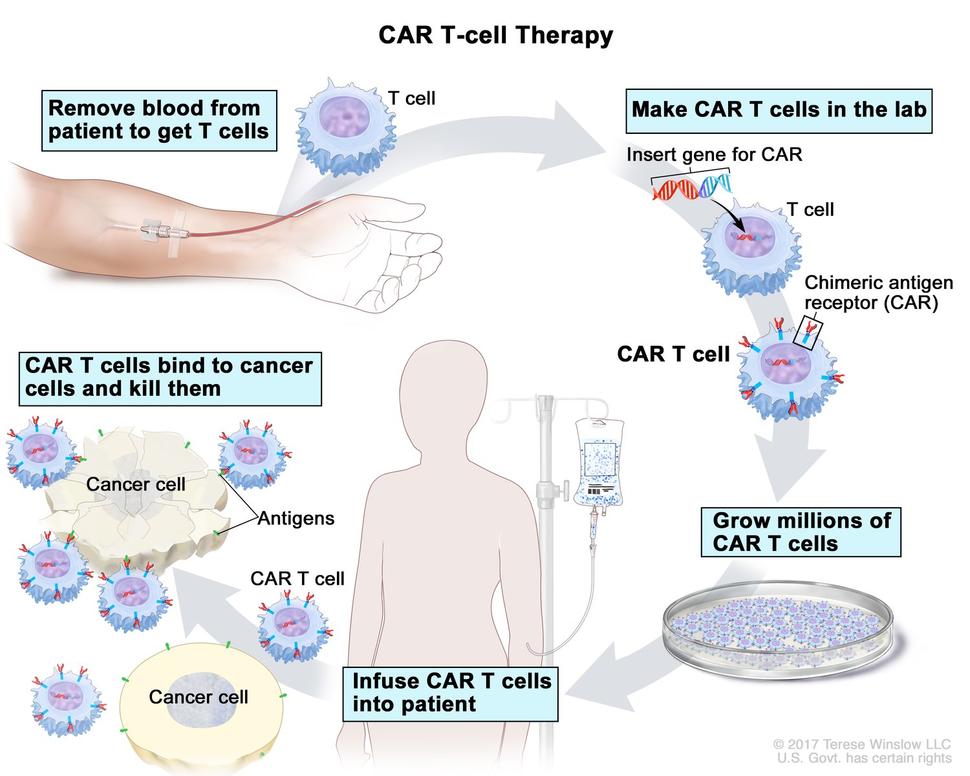
• Chimeric Antigen Receptor (CAR) T-cell therapy is a type of immunotherapy that uses a patient’s own T cells, a type of white blood cell, to recognize and attack cancer cells.
• In CAR T-cell therapy, T cells are taken from the patient’s blood and are changed in the lab by adding a gene for a receptor (called a chimeric antigen receptor or CAR), which helps the T cells attach to a specific cancer cell antigen. The CAR T cells are then given back to the patient.
• Since different cancers have different antigens, each CAR is made for a specific cancer’s antigen. For example, in certain kinds of leukemia or lymphoma, the cancer cells have an antigen called CD19. The CAR T-cell therapies to treat these cancers are made to that does not have the CD19 antigen.
• This type of treatment can be very helpful in treating some types of cancer, even when other treatments are no longer working..jpg)
Challenges:
• Preparation: The difficulty of preparing CAR T-cell therapies has been a major hindrance to their widespread use.
• Side Effects: In certain kinds of leukaemias and lymphomas, the efficacy is as high as 90%, whereas in other types of cancers it is significantly lower. The potential side-effects are also significant, associated with cytokine release syndrome (a widespread activation of
the immune system and collateral damage to the body’s normal cells) and neurological symptoms (severe confusion, seizures, and speech impairment).
• Affordability: Introduction of CAR T-cell therapy in India can face challenges of cost and value. Critics argue that developing CAR T-cell therapy in India may not be cost-effective as it will still be unaffordable for most people.
About T cells
• T cells, also known as T lymphocytes, are a type of white blood cell that plays a central role in the immune system. They are one of two primary types of lymphocytes, with B cells being the other type. T cells can be distinguished from other lymphocytes by the presence of a T-cell receptor (TCR) on their cell surface.

Address : 506, 3rd EYE THREE (III), Opp. Induben Khakhrawala, Girish Cold Drink Cross Road, CG Road, Navrangpura, Ahmedabad, 380009.
Mobile : 8469231587 / 9586028957
Telephone : 079-40098991
E-mail: dics.upsc@gmail.com
Address: A-306, The Landmark, Urjanagar-1, Opp. Spicy Street, Kudasan – Por Road, Kudasan, Gandhinagar – 382421
Mobile : 9723832444 / 9723932444
E-mail: dics.gnagar@gmail.com
Address: 2nd Floor, 9 Shivali Society, L&T Circle, opp. Ratri Bazar, Karelibaugh, Vadodara, 390018
Mobile : 9725692037 / 9725692054
E-mail: dics.vadodara@gmail.com
Address: 403, Raj Victoria, Opp. Pal Walkway, Near Galaxy Circle, Pal, Surat-394510
Mobile : 8401031583 / 8401031587
E-mail: dics.surat@gmail.com
Address: 303,305 K 158 Complex Above Magson, Sindhubhavan Road Ahmedabad-380059
Mobile : 9974751177 / 8469231587
E-mail: dicssbr@gmail.com
Address: 57/17, 2nd Floor, Old Rajinder Nagar Market, Bada Bazaar Marg, Delhi-60
Mobile : 9104830862 / 9104830865
E-mail: dics.newdelhi@gmail.com